日本Kikkoman食品分析-显色法检测组胺试剂盒
组胺是自体活性物质之一,在体内由组氨酸脱羧基而成,组织中的组胺是以无活性的结合型存在于肥大细胞和嗜碱性粒细胞的颗粒中,以皮肤、支气管粘膜、肠粘膜和神经系统中含量较多。
日本Kikkoman食品分析-显色法检测组胺试剂盒

组胺多数产生于金枪鱼,鲣鱼,鲭鱼此类海鲜的腐烂过程中。
对于以生鱼(鲜鱼、冷冻鱼肉)为原料的食品来说,为了防止组胺过量而导致过敏性中毒,对于食品组胺浓度测定的管理是很重要的。
但是,以前的组胺检测需要耗时一天,检测费用高,需要使用大型检测设备。如果用显色法组胺检测系统,生鱼(鲜鱼、冷冻鱼肉)的组胺含量检测可以快捷地完成。
获奖情况:
2007年日本水产学会的水产技术奖
2014年日本酱油协会的日本酱油技术奖(应用部门)
◆特点
● 简单: 前处理操作简单,可以省略荧光光谱法和高效液相色谱法那样测定障碍物质复杂的前处理操作。由于酶的特异性高,无需使用强酸或有机溶剂,任何对象均可简单、安全地分析
● 迅速: 利用具有高组胺特异性的组胺脱氢酶的酶法来检测。如果是生鱼,检测和提取过程仅需要1个小时即可得到结果。
● 廉价: 用廉价的试剂和检测器就可检测,免除了仪器成本。
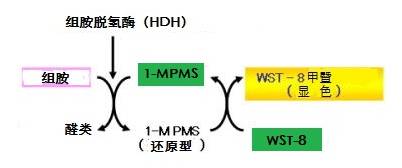
◆测定原理
组胺含有1-Methoxy PMS的情况下,添加组胺脱氢酶会被特异性分解。此时因产生的电子转移会通过四唑盐显色,便可以在470nm上下测量吸光度,计算组胺浓度。

◆性能
● 特异性:即使样本中含有尸胺和腐胺等其他的胺类也不影响检测结果。
● 测定范围:0.4-6ppm生鱼样本换算10-150ppm(稀释25倍)。
● 测定时间:各试剂注入后约20分钟。
◆试剂盒内容
|
试剂名
|
主要成分
|
包装
|
|
酶
|
组胺脱氢酶
|
冷冻干燥品 6瓶
|
|
显色试剂
|
WST-8/1-Methoxy PMS
|
冷冻干燥品 6瓶
|
|
缓冲液
|
Tris缓冲液
|
24mL 3瓶
|
|
标准液(30mL)
|
组胺
|
30mL 1瓶
|
(60次测定:10次*6)(2-8℃保存)
注意:
● 测定仪器请使用分光光度计(可检测到470nm左右) 。 *如需仪器推荐:分光光度计B
● 此试剂除了自主卫生检查和研究用途以外不可使用。
◆测定方法
● 下文所示显色法测定组胺实验手册
● 生鱼(分光光度计测定)
◆显色法测定组胺实验手册
实验器具准备
|
● 分光光度计
● 微量吸管
● 移液器(分注0.5mL溶液)
● 测量吸管
● 秒表
● 塑料试管2钟(容量约50mL、10mL)
● 试管架
● 水浴锅(设置到37℃)
|
● 遮光用的锡箔纸和箱子
● 天平(可测定1g类型)
● 粉碎机(研钵、匀浆器等)
● 漏斗
● 滤纸
● 旋涡混合器
● 冰浴装置
● 药匙
|
注意:组胺易吸附在玻璃上,试管等请使用塑料材质。
实验试剂准备
1、蒸馏水或者去离子水
2、0.1M EDTA·钠水溶液(pH=8):提取用溶液(将市售的0.5M EDTA溶液(pH=8)制备成5倍稀释液,可作为提取用溶液使用。)称取EDTA二钠二水合物37.2克,倒入1L容器。加入约750mL蒸馏水。搅拌混匀。用氢氧化钾水溶液或者NaOH水溶液调节pH值到8.0。pH值是在搅拌停止时的状态测定。将配置好的溶液倒入1L量筒,用蒸馏水定容到1L。制备完毕后倒入新容器,在室温下保存。
检测程序
|
[1]匀浆液制备
|
[2]提取
|
[3]过滤
|
 |
 |
 |
|
将鱼肉剁碎。选取1g碎鱼肉,添加24mL提取用溶液,搅拌均匀。
|
沸水浴20分钟加热处理。然后用冷水等冷却。(若是金枪鱼罐头,鱼粉的情况下不需要进行加热操作)
|
滤纸过滤
|
|
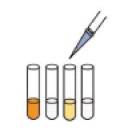
[4]试剂准备
|
[5]反应
|
[6]测定
|
 |
 |
 |
|
显色试剂,酶加水溶解。
|
将检测液以及各种试剂向试管中
分别注入0.5mL,遮光37℃孵育
15分钟。
|
测定吸光度。
|
操作流程
1.检测液的配制(注意:如果是熟鱼或者鱼罐头可省略④步骤。)
①取约10g样本(生鱼)置于粉碎机制成粉末。
②称量1g样本装入塑料试管(50mL容量)。
③向试管中加入24mL提取用溶液,用漩涡混合器拌匀。
④将试管置于沸水20分钟,加入提取。
⑤将试管置于冰上冷却后,清理样本碎块,用漩涡混合器拌匀。
⑥用滤纸(No.5C)过滤步骤⑤的提取液。
⑦抽取1mL或以上的滤液作为检测液。
2.试剂的准备
①用11mL蒸馏水溶解显色试剂(紫色标签)。
②用6mL缓冲液(粉红色标签)溶解酶试剂(绿色标签)。

3.测定
①准备4个塑料试管,如下表所示加入每种试剂。
(1次测定情况)同一样本3次测定的情况:(样本,空白样本,标准液,显色对照)*3=12
②遮光处理,37℃孵育15分钟,然后用分光光度计测定各反应液的吸光度。
| |
检测液Es
|
检测液空白Eb
|
标准液Estd
|
显色对照Ec
|
|
检测液(mL)
|
0.5
|
0.5
|
—
|
—
|
|
水(mL)
|
—
|
—
|
—
|
0.5
|
|
标准液(mL)
|
—
|
—
|
0.5
|
—
|
|
显色试剂(mL)
|
0.5
|
0.5
|
0.5
|
0.5
|
|
缓冲液(mL)
|
—
|
0.5
|
—
|
—
|
|
酶溶液(mL)
|
0.5
|
—
|
0.5
|
—
|
Es:检测液的吸光度 Eb:检测液空白的吸光度 Estd:标准液的吸光度 Ec:发光对照的吸光度
组胺浓度的计算方法
计算式:
样本液中的组胺含量(mg/L=ppm)=(Es-Eb)/(Estd-Ec)*4※1*25※2
样本液中的组胺含量(mg/L=ppm)=(Es-Eb)/(Estd-Ec)*100
※1 4:标准液的组胺浓度 ※2 25:样本溶液稀释25倍
其他组织样品的组胺检测试剂盒(欲了解详情请点击文字)
| 产品编号 |
产品名称 |
产品规格 |
产品等级 |
产品价格 |
| 308-16121 |
组氨检测试剂盒Histamine Test |
60次 |
- |
- |
















